How to read the data on hydroxychloroquine
More on the coronavirus drug debate. Is a 91.7% success rate with hydroxychloroquine and azithromycin — too good to be true? Dr. Kenny Lin, a otolaryngologist, offers a critical review of the latest data.
The Great Hydroxychloroquine Debate
A 91.7% success rate with hydroxychloroquine and azithromycin — too good to be true? A critical review of the latest data.

Hydroxychloroquine is the latest buzzword in the COVID-19 discourse. As I wrote previously, finding an effective treatment could significantly alter the trajectory of our global response to the pandemic. At his daily briefing on Sunday, Apr. 5, President Trump asked the country, “What do you have to lose?” and has been pushing for patient access to the drug through right-to-try legislation.
Hydroxychloroquine has been shown in the laboratory to be effective against coronaviruses including SARS and COVID-19 by blocking entry of the virus into cells. What works in a test tube, however, doesn’t always work in humans. In the case of SARS, hydroxychloroquine failed to lower viral loads in mice. Moreover, patients who routinely take hydroxychloroquine for lupus or arthritis have been infected with COVID-19 — showing that it is not preventative. Nevertheless, hydroxychloroquine may be a promising treatment and is one of many therapeutics that merits study.
I am personally hopeful that hydroxychloroquine, possibly combination with azithromycin or zinc, will be proven effective in clinical trials currently underway. We all want something to work. The caution, however, is that hydroxychloroquine, especially when combined with azithromycin, can be associated with visual and cardiac complications including arrhythmias. All physicians take the Hippocratic Oath in medical school — “do no harm” — and rightly hesitate to prescribe medications with known side effects in the absence of rigorous studies. Indeed, hospitals in France and Sweden have recently halted clinical trials with hydroxychloroquine citing increased cardiac risks with lack of benefit.
For daily updates on the latest hydroxychloroquine data, I suggest this blog by pharmacologist Derek Lowe, PhD. The existing data is, in a word, inconclusive. The majority of articles are still in “pre-print” — preliminary submissions to journals that may not have completed the peer review process required for formal publication. One recent pre-publication abstract and data table from Dr. Didier Raoult caught my attention and was also picked up by the New York Times and Fox News.
Briefly, Dr. Raoult has become a controversial figure who is championing the use of hydroxychloroquine with azithromycin (HCQ-AZ) to treat COVID-19. He initially published on Mar. 20 positive results in a group of 42 patients, of which 26 patients took HCQ-AZ. This paper was immediately criticized for its methodology including the fact that Dr. Raoult chose who received HCQ-AZ versus placebo, and analysis which excluded any patients who fared poorly on the regimen. A re-analysis of the data showed no benefit when accounting for the methodological inconsistencies. An official statement from the journal — the International Society of Antimicrobial Chemotherapy — stated the following: “The ISAC believes the article does not meet the Society’s expected standard, especially relating to the lack of better explanations of the inclusion criteria and the triage of patients to ensure patient safety.” A second publication by Dr. Raoult was issued as a pre-print without peer review and did not include a control group at all.
Nevertheless, the initial study sparked President Trump’s interest in hydroxychloroquine — he tweeted about it on Mar. 21 and has referred to it several times during his daily briefings.
The Study
On Apr. 10, Dr. Raoult posted to his Twitter a pre-publication abstract and data table summarizing his findings in just over 1,000 patients. Below, I will report the relevant findings (in bold) followed by the methodological issues and omissions that need to be answered.
- 1,061 out of 3,165 patients diagnosed with COVID-19 by PCR met inclusion criteria for this study. What are the inclusion criteria? Why were the other 2,000 patients excluded? Of note, there is no control group in this study as well. A gold standard clinical trial is randomized and double-blinded — meaning both the patient and investigator do not know who is in which group — so that the true effect of a treatment in matched groups can be assessed. In any study without a control, we cannot determine whether the observed outcomes are simply the result of the group of patients selected for inclusion in the study.
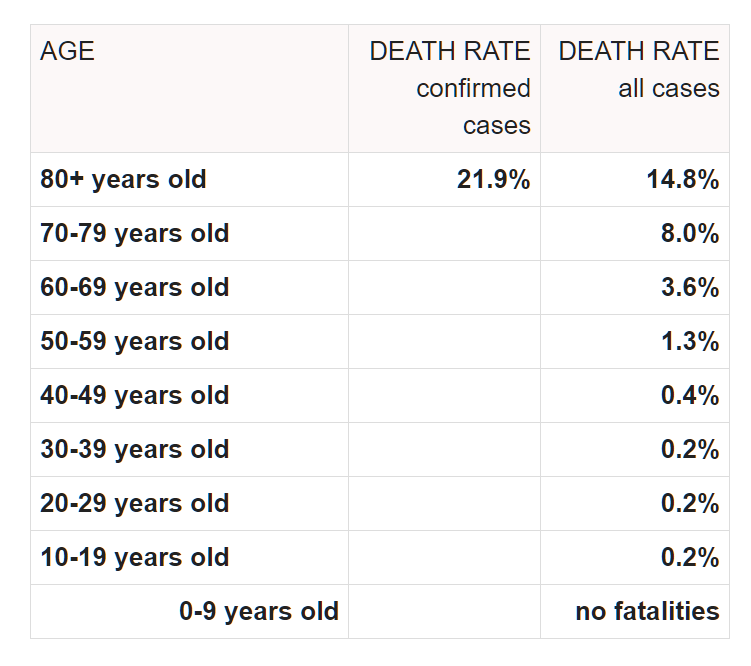
- Mean age of 43.6 years with standard deviation of 15.6. Dr. Raoult later writes in his conclusion that HCQ-AZ is a safe and effective treatment in elderly patients. Except — the math on the standard deviation tells me than 84% of patients were under the age of 59.2. Worldwide, current data suggests that expected mortality for patients under 60 ranges from 0.2% in younger patients to a high of 1.3% in patients 50–59 years. In other words, his study group was mostly younger patients who would be expected to have excellent outcomes irregardless of treatment. A comparison statistic for average age of hospitalized COVID-19 patient in France is difficult to find. In the United States, the average age of hospitalized patients is estimated to be in the 50s. Again, this would suggest that his study group is selected to be younger and healthier than average.
- A “good clinical outcome and virological cure was obtained in 973 patients within 10 days (91.7%). A good clinical outcome was defined clearance of viral shedding by day 10, and avoidance of hospitalization > 10 days, ICU transfer, or death. Additionally, it is noted that 4.4% of patients had prolonged viral carriage beyond day 10 (detectable on PCR) but negative viral culture; all but one cleared by PCR by day 15.
- Poor outcome in 46 patients (4.3%). This was defined as hospitalization > 10 days (31 patients), ICU transfer (10 patients), or death (5 patients). Of note, the 5 patients that died were age 74–95. Of the 31 hospitalized > 10 days, 16 are still hospitalized at the time of publication. Overall the death rate was 0.5% with another 16 (1.6%) of patients in the hospital.
- Poorer outcomes associated with older age, worse initial severity, low HCQ serum concentration. Also found worse outcomes associated with patients who take beta-blockers and angiotension II receptor blockers. These findings have been reported elsewhere and are in agreement with the global clinical experience.
- Mortality for patients with >3 days of HCQ-AZ therapy is lower than patients treated with other regimens at the same hospital (IHU) and within the Marseille public hospitals. This statement is not validated with data showing the outcomes with other regimens and at Marseille public hospitals.
- In the data table, the authors additionally remark that data was only available for 928 patients. The initial reported n of 1,061 is misleading as 56 patients were excluded for being asymptomatic and 77 patients had missing data.
Conclusion
Dr. Raoult’s latest publication appears very promising on first glance — 91.7% cure within 10 days and 0.5% mortality! However, I am concerned about the lack of a control group and the lack of transparency about the patient selection process. An intentional or unintentional selection of a younger and healthier patient cohort may result in excellent outcomes irregardless of hydroxychloroquine. We need the full manuscript to evaluate whether this abstract is indeed promising or misleading.
The gold standard remains a double-blind, randomized control trial. I sincerely hope we get data from such trials in the near future.